Revision Notes of Acids, Bases and Salts?
Class 10th – Science (Chemistry)
Chapter-2
Acids, Bases and Salts
Topics in the Chapter
- Introduction
• Properties of Acids
• Properties of Bases
• Types of Indicators and its properties
• Reaction of Acids and Bases with Metals
• Reaction of Acids with Metal Carbonates and Metal Hydrogen carbonates
• Reaction of Acids and Bases with each other
• Reaction of Metallic Oxides with Acids
• Reaction of a Non-metallic Oxide with Base
• Similarities between all Acids and all Bases
• Acid or Base in Water Solution
• Strength of Acids and Base solutions - Types of salts
- Some Important salts
INTRODUCTION
- Have you ever tasted lemon without adding water and salt? Or have you noticed the slippery touch of soap?
- Lemon juice, tamarind, raw mango, etc. taste sour, some substances like baking powder tastes bitter. Common salt taste salty.
- These tastes are the characteristics of three different types of compounds that are acids, bases and salts.
ACIDS
- The chemical nature of acids is acidic. The word acid comes from Latin word acere which means sour.
- Taste of substances containing acids is sour e.ge curd, lemon juice, orange juice, vinegar, etc.
- Robert Boyle was the first to study the properties of acids and describe them is sour, corrosive and turning blue litmus red.
| Name of Acid |
Found in |
| Acetic acid
Formic acid Citric acid Lactic acid Oxalic acid Ascorbic acid (vitamin ‘C’) Tartaric acid |
Vinegar
Ant’s sting Lemon, orange Curd Spinach Amla Grapes, Tamarind, Ripe mango |
Preparation of Acids
Acids are formed when non metallic oxides are dissolved in water.
SO2 + H2O → H2SO4 Sulphur dioxide Water Sulphuric acid
CO2 + H2O → H2CO3 Carbon dioxide Water Carbonic acid
Properties of Acids
- They are sour to taste
- They are corrosive. They destroy clothes, skin and paper.
- Acids turn blue litmus to red.
- Acids turn methyl orange solution to reddish pink.
- All of them contain hydrogen ions H+
- Acids react with bases to give salt and water.
- The PH value of acid is bellow 7.
Basicity of acids
- The number of replaceable hydrogen atoms in a molecule of an acid is called its Basicity.
Uses of Acids
- Sulphuric acid and nitric acid are used in manufacture of fertilizers, paints, explosives, etc.
- Sulphuric acid is used in the batteries for cars, inverters, etc.
- Acetic acid is used in preservation of food and for enhancing flavour of food.
- Tartaric acid is used in making baking powder by mixing it with baking soda.
- Our body produces hydrochloric acid in stomach. This acid makes it possible for the human.
- Aqua regia is very powerful acid. It can dissolve gold. It is made of one part nitric acid and three parts of concentrated hydrochloric acid.
ACID RAIN
- The rain containing excess of acids is called acid rain.
- Rain becomes acidic because carbon dioxide, sulphur dioxide and nitrogen dioxide which are released into air as pollutants dissolve in rain drops to form carbonic acid, sulphuric acid and nitric acid respectively.
- Acid rain damages buildings, historical monuments, plants and animals.
BASES
Substances which are soapy to touch and are bitter in taste are called bases, eg. Baking soda, washing soda, calcium hydroxide etc.
Blood when healthy is basic in nature Saliva in our mouth also basic.
Preparation of Bases
When metals burn in oxygen the respective metallic oxides are formed.
The oxide when dissolved in water give the respective hydroxide.
Na2O + H2O → NaOH Sodium oxide Water Sodium hydroxide
K2O + H2O → KOH Potassium oxide Water Potassium hydroxide
But there are other metallic hydroxides like Cupric hydroxide, Ferric hydroxide, etc. These cannot be obtained by the above method.
Properties of Bases :
- Bases are bitter to taste.
- They are soupy to touch.
- All bases turn red litmus to blue.
- Bases turn colourless phenolphthalein to pink and orange coloured methyl orange indicator to yellow.
- Alkali reacts with ammonium salts to liberate ammonia.
- PH value of bases is >7
Weak and Strong Bases
Strong bases:
Some bases are readily soluble in water. These are strong bases. These are also called alkalies for example: Sodium hydroxide and Potassium hydroxide.
Weak bases:
Some bases are insoluble or partly soluble in water such bases are called weak base. For example: Ammonium hydroxide, Calcium hydroxide.
NOTE: Strong bases are very corrosive and can burn the skin.
Uses of Bases
- Calcium hydroxide is used as an ingredient in whitewash, neutralizing acidic soil, in making bleaching powder and softening hard water.
- Magnesium hydroxide also known as milk of magnesia is used as an antacids.
- Sodium hydroxide also known as caustic soda is used in manufacturing of soaps, paper and textile.
- Ammonium hydroxide is used in household cleaners and in fertilizers.
- Sodium carbonate along with sulphuric acid is used in fire extinguisher.
Indicators:
Substances which change their colour/smell in different types of substances (like acids and bases) to show the acidic and basic behavior.
Types of Indicators:
(i) Natural indicators
(ii) Synthetic indicators
(iii) Olfactory indicators (iv) Universal indicators
(i) Natural indicators: Found in nature in plants. Examples: Litmus, red cabbage leaves extract, flowers of hydrangea plant, turmeric.
(ii) Synthetic indicators: These are chemical substances. Examples: Methyl orange, phenolphthalein.
(iii) Olfactory indicators: These substances have different odour in acid and bases.
Chemical properties of Acids and Bases
→ Reaction of Acids with Metals
• Acids react with metal to form metallic salt and releases Hydrogen Gas.
Acid + Metal → Salt + Hydrogen Gas
• Example: Zinc granules react with dilute Hydrochloric acid in a test tube.
2HCl + Zn → ZnCl2 + H2
→ Reaction of Bases with Metals
- Bases react with metal to evolve hydrogen Gas. Also, note that all metals do not react with bases. The metal must be more reactive than the metals present in the base for the reaction to take place.
Base + Metal → Salt + Hydrogen gas• Example: Zinc granules react with NaOH solution to form sodium zincate and evolve hydrogen gas.
2NaOH + Zn → Na2ZnO2+ H2• Hydrogen gas released can be tested by bringing burning candle near gas bubbles, it burst with pop sound.Reaction of Acids with Metal Carbonates and Metal bicarbonates• Acids reacts with Metal Carbonates and Metal Hydrogen carbonates to form Salt, Carbon dioxide and water.
Metal carbonate/bicarbonate + Acid → Salt + Carbon dioxide + Water - Examples:
(i) 2HCl + Na2CO3 → 2NaCl + CO2 + H2O
(ii) HCl + NaHCO3 → NaCl + CO2 + H2O
- CO2can be tested by passing it through lime water. It turns lime water milky.
Ca(OH)2 + CO2 → CaCO3 + H2O
- When excess CO2 is passed, milkiness disappears.
CaCO3 + CO2 + H2O → Ca(HCO)3
Reaction of Acids and Bases with each other
• Acids and Bases react to form salt and water.
Acid + Base → Salt + H2O
• Neutralisation Reaction: Reaction of acid with a base is called as neutralization reaction.
Example: HCl + NaOH → NaCl + H2O
• Strong Acid + Weak Base → Acidic salt + H2O
• Weak Acid + Strong Base → Basic salt + H2O
• Strong Acid + Strong Base → Neutral salt + H2O
• Weak Acid + Weak Base → Neutral salt + H2O
Reaction of Metallic Oxides with Acids
→ Metallic oxides are basic in nature.
Example: CaO, MgO are basic oxides.
Metallic Oxide + Acid → Salt + H2O
CaO + 2HCl → CaCl2 + H2O
Reaction of Non-metallic Oxides with Bases
→ Non-metallic oxides are acidic in nature.
→ Non-metallic Oxide + Base → Salt + H2O
CO2 + Ca(OH)2 → CaCO3 + H2O
Reaction of Acid
(i) Acid + Metal Carbonate → Salt + CO2 + Water
(ii) Acid + Metal → Salt + H2
(iii) Acid + Metal Hydrogen Carbonate → Salt + CO2 + H2O
(iv) Acid + Metallic oxide → Salt +H2O
(v) Acid + Base → Salt + H2O
Reaction Of Base
(i) Base + Metal → Salt + H2
(ii) Base + Metal Carbonate → No Reaction
(iii) Base + Metal Hydrogen Carbonate → No Reaction
(iv) Base + Acid → Salt + H2O
(v) Base + Non Metallic oxide → Salt + H2O
Similarities between all Acids and all Bases
→ All acids have H+ ions in common. All acids produce H+ ions
→ Acids produce H+ ions in solution which are responsible for their acidic properties.
→ All bases have OH– (hydroxyl ions) in common. All bases produce OH– ions
Acid or Base in Water Solution
→ Acids produce H+ ions in presence of water.
→ H+ ions cannot exist alone, they exist as H3O+ (hydronium ions).
H+ + H2O → H3O+
HCl + H2O → H3O+ + Cl–
→ Bases when dissolved in water gives OH− ions.
NaOH → Na+ + OH–
Mg (OH)2 + H2O → Mg2- + 2OH–
→ Bases soluble in water are called alkali.
→ While diluting acids, it is recommended that the acid should be added to water and not water to acid because the process of dissolving a acid or a base in water is highly exothermic.
Strength of Acids and Base solutions
→ Strength of acid or base can be estimated using universal indicator.
→ Universal indicator: It is a mixture of several indicators. It shows different colours at different concentrations of H+ ions in the solution.
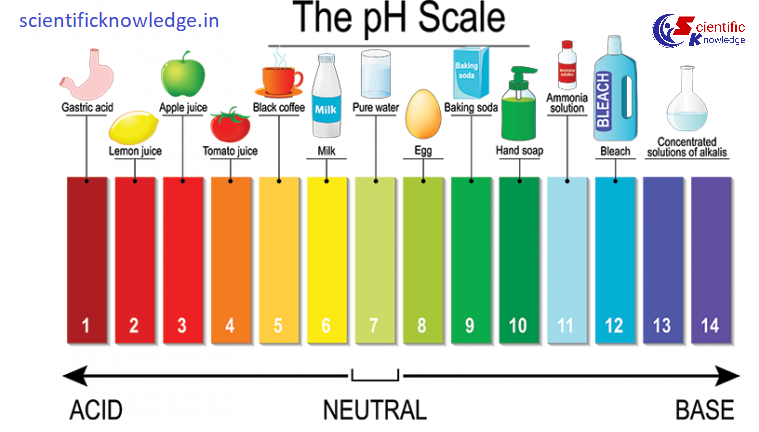
→ pH Scale: A scale for measuring H+ ion concentration in a solution. p in pH stands for ‘potenz’ a German word which means power. Neutral solutions (such as pure water) have a pH of 7, acidic solutions have a pH lower than 7, and alkaline solutions have a pH higher than 7.
Salts:
Salts are the ionic compounds which are produced after the neutralization reaction between acid and base. Salts are electrically neutral. There are number of salts but sodium chloride is the most common among them. Sodium chloride is also known as table salt or common salt. Sodium chloride is used to enhance the taste of food.
Characteristics of salt:
- Most of the salts are crystalline soild.
- Salts may be transparent or opaque.
- Most of the salts are soluble in water.
- Solution of the salts conducts electricity in their molten state also.
- The salt may be salty, sour, sweet, bitter and umami (savoury).
- Neutral salts are odourless.
- Salts can be colourless or coloured.
Family of Salt: Salts having common acidic or basic radicals are said to belong to the same family.
Example:
(i) Sodium chloride (NaCl) and Calcium chloride (CaCl2) belongs to chloride family.
(ii) Calcium chloride (CaCl2) and Calcium sulphate (CaSO4) belongs to calcium family.
(iii) Zinc chloride (ZnCl2) and Zinc sulphate (ZnSO4) belongs to the zinc family.
Classification of salts:
Based on nature the salts have been classified into different types. They are:
- Neutral salts
- Acidic salts
- Basic salts
- Double salts
- Complex salts
(i) Neutral Salt: Salts produced because of reaction between a strong acid and strong base are neutral in nature. The pH value of such salts is equal to 7, i.e. neutral.
Example : Sodium chloride, Sodium sulphate. Postassium chloride, etc.
Sodium chloride (NaCl): It is formed after the reaction between hydrochloric acid (a strong acid) and sodium hydroxide (a strong base).
NaOH (aq) + HCl (aq) → NaCl (aq) + H2O (l)
Sodium Hydroxide Hydrochloric acid Sodium chloride Water
Sodium Sulphate (Na2SO4): It is formed after the reaction between sodium hydroxide (a strong base) and sulphuric acid ( a strong acid).
2NaOH (aq) + H2SO4 (aq) → Na2SO4 (aq) + 2H2O (l)
Sodium Hydroxide Sulphuric acid Sodium Sulphate Water
Potassium Chloride (KCl): It is formed after the reaction between potassium hydroxide (a strong base) and hydrochloric acid (a strong acid).
KOH (aq) + HCl (aq) → KCl (aq) + H2O (l)
Potassium Hydroxide Hydrochloric Acid Potassium chloride Water
(ii) Acidic Salts:
Salts which are formed after the reaction between a strong acid and weak base are called Acidic salts. The pH value of acidic salt is lower than 7. For example Ammonium sulphate, Ammonium chloride, etc.
Ammonium chloride is formed after reaction between hydrochloric acid (a strong acid) and ammonium hydroxide (a weak base).
NH4OH (aq) + HCl (aq) → NH4Cl (aq) + H2O (l)
Ammonium Hydroxide Hydrochloric Acid Ammonium chloride Water
Ammonium sulphate is formed after reaction between ammonium hydroxide (a weak base) and sulphuric acid (a strong acid).
NH4OH (aq) + H2SO4 (aq) → (NH4)2SO4 (aq) + H2O (l)
Ammonium Hydroxide Sulphuric Acid Ammonium Sulphate Water
(iii) Basic Salts:
Salts which are formed after the reaction between a weak acid and strong base are called Basic Salts. For example; Sodium carbonate, Sodium acetate, etc.
H2CO3 (aq) + 2NaOH (aq) → Na2CO3 + H2O (l)
Carbonic Acid Sodium Hydroxide Sodium Carbonate Water
Sodium acetate is formed after the reaction between a strong base, sodium hydroxide (a strong base) and acetic acid, (a weak acid).
CH3COOH (aq) + 2NaOH (aq) → CH3COONa + H2O (l)
Acetic Acid Sodium Hydroxide Sodium Acetate Water
Cause of formation of acidic, basic and neutral salts:
- When a strong acid reacts with a weak base, the base is unable to fully neutralize the acid. Due to this, an acidic salt is formed.
- When a strong base reacts with a weak acid, the acid is unable to fully neutralize the base. Due to this, a basic salt is formed.
- When equally strong acid and a base react, they fully neutralize each other. Due to this, a neutral salt is formed.
pH value of salt:
- Neutral salt: The pH value of a neutral salt is almost equal to 7.
- Acidic salt: The pH value of an acidic salt is less than 7.
- Basic salt: The pH value of a basic salt is more than 7.

Some Important Salts
- Common Salt (Sodium Chloride):Sodium chloride (NaCl) is also known as Common or Table Salt. It is formed after the reaction between sodium hydroxide and hydrochloric acid. It is a neutral salt. The pH value of sodium chloride is about 7. Sodium chloride is used to enhance the taste of food. Sodium chloride is used in the manufacturing of many chemicals.
Important chemical from sodium chloride
Sodium Hydroxide (NaOH):
Sodium hydroxide is a strong base. It is also known as caustic soda. It is obtained by the electrolytic decomposition of solution of sodium chloride (brine). In the process of electrolytic decomposition of brine (aqueous solution of sodium chloride), brine decomposes to form sodium hydroxide. In this process, chlorine is obtained at anode and hydrogen gas is obtained at cathode as by products. This whole process is known as Chlor – Alkali process.
2NaCl (aq) + 2H2O (l) → 2NaOH (aq) + Cl2 + H2 (g)
Sodium chloried Water Sodium Hydroxide Chlorine Hydrogen
Use of products after the electrolysis of brine:
Hydrogen gas is used as fuel, margarine, in making of ammonia for fertilizer, etc.
- Chlorine gas is used in water treatment, manufacturing of PVC, disinfectants, CFC, pesticides. It is also used in the manufacturing of bleaching powder and hydrochloric acid.
- Sodium hydroxide is used for degreasing of metals, manufacturing of paper, soap, detergents, artificial fibres, bleach, etc.
- Bleaching Powder (CaOCl2):
Bleaching powder is also known as chloride of lime. It is a solid and yellowish white in colour. Bleaching powder can be easily identified by the strong smell of chlorine.
When calcium hydroxide (slaked lime) reacts with chlorine, it gives calcium oxychloride (bleaching powder) and water is formed.
Ca(OH)2 (aq) + Cl2 (aq) → CaOCl2 (aq) + H2O (l)
Slaked lime Chlorine Bleaching powder Water
Aqueous solution of bleaching powder is basic in nature. The term bleach means removal of colour. Bleaching powder is often used as bleaching agent. It works because of oxidation. Chlorine in the bleaching powder is responsible for bleaching effect.
Use of Bleaching Powder:
- Bleaching powder is used as disinfectant to clean water, moss remover, weed killers, etc.
- Bleaching powder is used for bleaching of cotton in textile industry, bleaching of wood pulp in paper industry.
- Bleaching powder is used as oxidizing agent in many industries, such as textiles industry, paper industry, etc.
- Baking Soda (NaHCO3):Baking soda is another important product which can be obtained using byproducts of chlor – alkali process. The chemical name of baking soda is sodium hydrogen carbonate (NaHCO3) or sodium bicarbonate. Bread soda, cooking soda, bicarbonate of soda, sodium bicarb, bicarb of soda or simply bicarb, etc. are some other names of baking soda.
Preparation Method: Baking soda is obtained by the reaction of brine with carbon dioxide and ammonia. This is known as Solvay process.
NaCl + H2O + CO2 + NH3 → NH4Cl + NaHCO3 Sodium chloride Water Carbon dioxide Ammonia Ammonium chloride Sodium bicarbonate
In this process, calcium carbonate is used as the source of CO2 and the resultant calcium oxide is used to recover ammonia from ammonium chloride.
Properties of Sodium Bicarbonate:
- Sodium bicarbonate is white crystalline solid, but it appears as fine powder.
- Sodium hydrogen carbonate is amphoteric in nature.
- Sodium hydrogen carbonate is sparingly soluble in water.
- Thermal decomposition of sodium hydrogen carbonate (baking soda).
- When baking soda is heated, it decomposes into sodium carbonate, carbon dioxide and water.
2NaHCO3 + heat → Na2CO3 + CO2 + H2O - Sodium carbonate formed after thermal decomposition of sodium hydrogen carbonate decomposes into sodium oxide and carbon dioxide on further heating.
Na2CO3 → Na2O + CO2
This reaction is known as Dehydration reaction.
Use of Baking Soda:
- Baking soda is used in making of baking powder, which is used in cooking as it produces carbon dioxide which makes the batter soft and spongy.
- Baking soda is used as an antacid.
- Baking soda is used in toothpaste which makes the teeth white and plaque free.
- Baking soda is used in cleansing of ornaments made of silver.
- Since sodium hydrogen carbonate gives carbon dioxide and sodium oxide on strong heating, thus, it, is used as a fire extinguisher.
Baking Powder:
Baking powder produces carbon dioxide on heating, so it is used in cooking to make the batter spongy. Although, baking soda also produces carbon dioxide on heating, but it is not used in cooking because on heating, baking soda produces sodium carbonate along with carbon dioxide. The sodium carbonate, thus, produced, makes the taste bitter.
NaHCO3 + Heat → Na2CO3 + CO2 + H2O (l)
Baking soda Sodium carbonate Carbon dioxide Water
Baking powder is the mixture of baking soda and a mild edible acid. Generally, tartaric acid is mixed with baking soda to make baking powder.
NaHCO3 + C4H6O6 → Na2C4H6O6 + CO2 + H2O (l)
Baking soda Tartaric acid Sodium tartrate Carbon dioxide Water
When baking powder is heated, sodium hydrogen carbonate (NaHCO3) decomposes to give CO2 and sodium carbonate (Na2CO3). CO2 causes bread and cake fluffy. Tartaric acid helps to remove bitter taste due to formation of Na2CO3.
- Washing Soda (Sodium Carbonate)
Preparation Method:
Sodium carbonate is manufactured by the thermal decomposition of sodium hydrogen carbonate obtained by Solvay process.
NaCl + H2O + CO2 + NH3 → NH4Cl + NaHCO3 Sodium chloride Water Carbon dioxide Ammonia Ammonium chloride Sodium bicarbonate
NaHCO3 + Heat → Na2CO3 + CO2 + H2O (l) Sodium bicarbonate Sodium carbonate Carbon dioxide Water
The sodium carbonate obtained in this process is dry. It is called Soda ash or Anhydrous sodium carbonate. Washing soda is obtained by rehydration of anhydrous sodium carbonate.
NaCO3 + 10 H2O → Na2CO3 . 10 H2O
Sodium carbonate Water Sodium carbonate(Hydrated)
Since there are 10 water molecules in washing soda, hence, it is known as Sodium Bicarbonate Decahydrate.
Sodium carbonate is a crystalline solid and it is soluble in water when most of the carbonates are insoluble in water.
Use of sodium carbonate:
- It is used in the cleaning of cloths, especially in rural areas.
- In the making of detergent cake and powder.
- In removing the permanent hardness of water.
- It is used in glass and paper industries.
- Plaster of paris (CaSO4 . H2O) :
Plaster of paris which is chemically called calcium sulphate hemihydrates.
Since it is brought to use from paris, called as “plaster of paris”.
It is prepared by heating of gypsum at 373K. CaSO4.2H2O + Heat (373 K) → CaSO4 .1/2 H2O + H2O
Gypsum Plaster of Paris WaterUses:
- It is used as a bandage, proofing material, sealing agent.
- It is used for making statues, toys and decorative articles.
- It is also used for smoothening wall surfaces.
- Double salts:
Salts that are formed by mixing of two simple salts which are obtained crystallisation.
Example:
Potash alum – K2SO4Al2(SO4)3 .24H2O
Dolomite – CaCO3.MgCO3 - Complex salts:
The salts which contains different types of metal atoms which on hydrolysis produces complex ions along with simple ions are called complex salts.
Example:
[Ag(NH3)2]Cl ⇄ [Ag ( NH3 )2 ]+ + Cl–
The water of Crystallization:
Many salts contain water molecule and are known as Hydrated Salts. The water molecule present in salt is known as Water of crystallization.
Examples:
Copper sulphate penta hydrate (CuSO4.5H2O): Blue colour of copper sulphate is due to presence of 5 molecules of water. When copper sulphate is heated, it loses water molecules and turns into grey – white colour, which is known as anhydrous copper sulphate. After adding water, anhydrous copper sulphate becomes blue again.
CuSO4 . 5H2O + Heat → CuSO4
Coppersulphate (Hydrated) Coppersulphate (anhydrous)
Some of the hydrated salts along with their chemical formula.
| Name of the salt | Chemical formula |
| Sodium carbonate decahydrate | Na2CO3.10 H2O |
| Zinc Sulphate heptahydrate or White vitriol | ZnSO4.7H2O |
| Magnesium sulphate heptahydrate or Epsom salt | MgSO4.7H2O |
| Potash alum | K2SO4 Al2 (SO4)3 .24H2O |
| Copper (II) sulphate pentahydrate or Blue vitriol | CuSO4.5H2O |
| Calcium sulphate dihydrate or Gypsum | CaSO4.2H2O |
Revision Notes of Chemical Reaction and Equations?
Revision Notes of Acids, Bases and Salts?




More Stories
Full Revision रासायनिक अभिक्रियाएँ एवं समीकरण
अंबेडकर नगर बौद्धिक पत्रक मार्च-अप्रैल 2023
10 Important Exam Tips